Description:
Background Preterm birth is the leading cause of death among children, accounting for 18% of all deaths among children aged under 5 years and as much as 35% of all deaths among newborns. Preterm birth due to preterm labor is a challenging concern for the practicing obstetrician and few drugs have been proven to be effective in the treatment of preterm labor. Unfortunately, tocolytic modalities that have shown to be effective are often limited in their use due to associated toxicities to the fetus. Innovative solutions for preterm labor management are in high demand.
Discovery/Highlights Investigators from the University of Texas Health Science Center at Houston (UTHealth), in collaboration with Houston Methodist Hospital (HMH), have developed a novel technology comprised of a delivery system for a tocolytic drug (indomethacin) targeted to the site of action (maternal uterus) to enhance the drug efficacy and prevent fetal toxicities. This invention focus on the development of uterus-targeted nanoparticles to ensure the drugs are delivered to the desired sites of action, thereby minimizing or eliminating off-target effects.
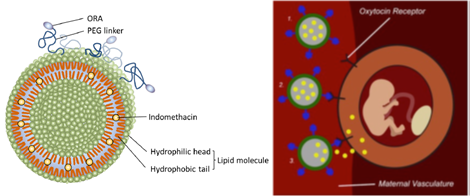
Clinical impact If the biodistribution of indomethacin (IND) could be restricted to the maternal compartment, specifically to the uterine myometrium, and not affect the fetus, then prolonged administration of IND could be effective in the treatment of preterm labor and positively impact neonatal morbidity associated with preterm birth. One in four women is hospitalized for preterm labor during pregnancy. Preterm birth represents 50% of all infant hospital costs per year. According to the March of Dimes, in the US a premature baby spends an average of 25.4 days in a specialty care nursery at an average cost of $144,692.
Stage of Development Pre-clinical
Intellectual Property Status
Licensing Availability Available for licensing worldwide.